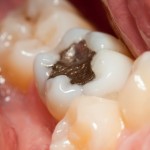
The FDA concluded in 2009 that dental amalgam was a safe and effective dental material and issued a final rule, which the American Dental Association (ADA) supported. The FDA categorizes encapsulated dental amalgam as a class II medical device, placing it in the same class as gold- and tooth-colored composite fillings. The ADA has supported a class II designation for dental amalgam since 2002, when the FDA first proposed it.
Dental amalgam is a cavity-filling material made by combining mercury with other metals such as silver, copper, and tin. Numerous scientific studies conducted over the past several decades, including two large clinical trials published in the April 2006 issue of the The Journal of the American Medical Association, indicate dental amalgam is a safe, effective, cavity-filling material. In its 2009 review of the scientific literature on amalgam safety, the ADA’s Council on Scientific Affairs reaffirmed that the scientific evidence continues to support amalgam as a safe choice for dental patients.





